In our previous blog “What do MDR & LIR legislation mean for you as a hospital, healthcare institution or manufacturer?” we already explained which information you as a hospital or manufacturer will have to process to comply with the new legislation. Do you still know which data you have to provide to the EPD, the LIR, or to the patient? Then take another look at that blog as a reminder. Now we go a step further and take you along in the process: how do you receive and send the right data for your organization to be MDR & LIR-proof?
The exchange of product data and the challenges herein
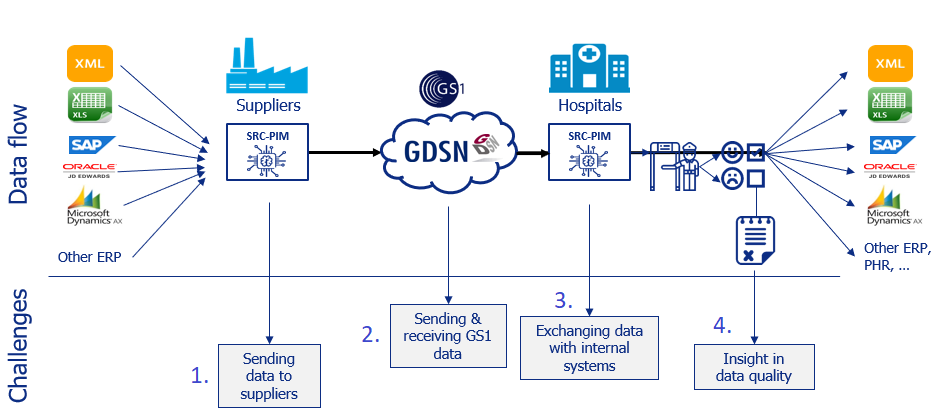
In this diagram, you’ll find the supplier on the left side, and on the right side, the hospital. In between is the GS1 data pool, where the product data is stored in a structured and standardized way. But how does a supplier (e.g. manufacturer) get the product data there, and how does a recipient (e.g. hospital) get the product data from the data pool and to your follow-up systems such as an ERP for example? And how item data quality checked? Let’s look at these four challenges.
Challenge 1: supplying supplier data
As a manufacturer, you will need to send product data to GS1 due to the LIR, MDR, and IVDR (In Vitro Diagnostic Regulation) legislations. Hospitals that manufacture their own medical devices, for example, with a 3D printer, must also comply. Many manufacturers ask us these questions: what information do I need to provide, in what format? These are a common challenges. And how does a manufacturer know if the information he provides suffices? Because the GS1 Healthcare data model is updated approximately every three months, it is therefore very difficult for suppliers of item data to keep track of which fields have been added, changed, or deleted. This is solved by SRC-PIM. Because SRC-PIM always includes the most recent data model in the software, the supplier of product data for medical devices will always be able to see what has changed in the data model, and what new or different information he has to provide. This avoids an enormously time-consuming process of sending erroneous data back and forth. The product data that you send to GS1 as a manufacturer or as a hospital will always be “First Time Right”.
Challenge 2: receiving GS1 data
To comply with LIR, MDR, and IVDR, the recipients of product data – usually hospitals or healthcare institutions – must be able to retrieve the product data from GDSN. To do this, hospitals must subscribe to a supplier’s ‘GLN’ (Global Location Number) before they can receive the data. Some hospitals may have thousands of suppliers, and by subscribing they will receive all articles and all product data. This causes a data pollution on an epic scale in their ERP systems, causing systems to fail and purchasers unable to see the forest for the tree. This needs to be improved.
Challenge 3: data interfacing with own systems
In order not to flood your own systems with item data that you do not need and do not use, SRC-PIM serves not only as a guard but also as a controller. Only the selection of articles you make yourself will be delivered in your own systems, but also only if the article information meets the validation rules of GS1. Moreover, you also get the opportunity to check and enrich the item data yourself, before sending it to your ERP, and further to the EPD or LIR. Thanks to the workflow functionality, it is also possible to make different departments in your hospital or healthcare institution responsible for handling certain information. In this way, you also keep your hospital GDPR-proof.
Challenge 4: making data quality transparent
Now you have all the desired item data in your own systems, but item data is not static and is constantly changing. So when item data is changed by a supplier, you as a recipient will receive a notification and get insight into the changed items. Only after your own check, the changed data will go be entered into your own system.
You can also delete items if you no longer use them, and you can also send a quality report to your supplier so that the data can be changed at the source. All this saves you not hours, but days.
SRC-PIM will thus act as your tool to filter, enrich, and check item data and to control your suppliers accordingly. After this, you can use these product data without any problem in your follow-up systems.
From IC to IT: unique offer, take advantage now!
So is your organization behind on getting your product data in order? Or do you not yet comply with the obligation to receive or send product data via GDSN, for example, if you, as a hospital, also produce medical devices yourself using a 3D printer?
Then we have a unique offer for you. Book our free workshop now, which normally costs € 1.879 and discover how to get your medical device item data in order. Get in touch with us via sales@src.nl or click through below this blog.
After a session with our expert, we will provide you with extensive advice and a clear plan of action tailored to your organization. So you can get off to a good start right away!